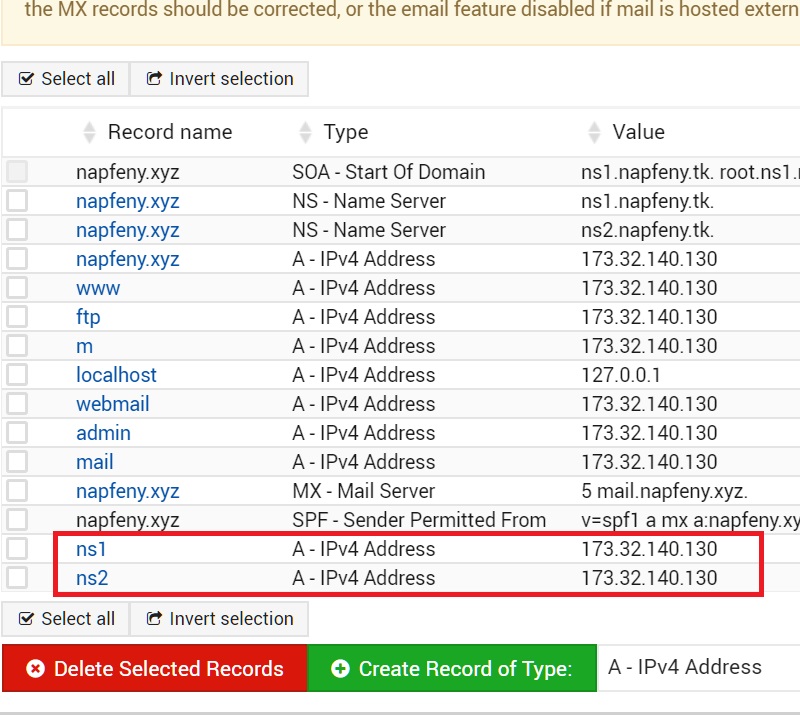
Yeast diploids co-expressing full length A/H1N1/Puerto Rico/8/34 virus NS1 protein fused to the Gal4 DNA-binding domain and full-length ADAR1 or ADAR1 truncated mutants fused to Gal4 activating domain were spotted onto a plate containing medium without histidine. Zα DNA-binding domain (grey boxes), dsRNA binding domain (DRBD, purple boxes), adenosine deaminase domain (yellow boxes). (B) Schematic representation of full-length and truncated ADAR1 proteins.
(A) Subcellular localization of ADAR1 (green), HA and NS1 (red) 8 h post infection of A549 cells. Vertical dashed lines indicate the mean betweenness value for each distribution. Solid lines represent linear regression fits. P(b) is the probability for a node to have a betweenness value of b in the network. (D) Betweenness distribution of human proteins and human proteins targeted by NS1 and/or NS2 proteins in the human interactome. Vertical dashed lines indicate the mean degree of each distribution. P(k) is the probability of a node to connect k other nodes in the network. (C) Degree distribution of human proteins and human proteins targeted by NS1 and/or NS2 proteins in the human interactome. (B) Frequency of interactions between individual host cell factors and NS1 and/or NS2 proteins of the 9 different influenza virus strains. DRBD-containing proteins (DRBPs) are indicated with a star. The 11 NS1 and the single NS2 interactors described earlier are highlighted with bold letters. The 33 NS1-specific interactors are indicated in blue, 28 NS2-specific interactors in grey and shared interactors in yellow. ADAR1 editing activity was enhanced by both viruses through dengue virus NS3 and influenza virus NS1 proteins, suggesting a similar virus-host co-evolution. Surprisingly, ADAR1 also appeared as a pro-viral host factor for dengue virus replication and directly interacted with the viral NS3 protein. In particular, adenosine deaminase acting on RNA 1 (ADAR1) appeared as a pro-viral host factor whose expression is necessary for optimal viral protein synthesis and replication. One of the main factors targeted by NS1 of all virus strains was double-stranded RNA binding domain protein family. Validation of the relevance of these host cell proteins for the viral replication cycle revealed that 7 of the 79 NS1 and/or NS2-interacting proteins positively or negatively controlled virus replication. These interacting proteins are potentially involved in each step of the infectious process and their contribution to viral replication was tested by RNA interference. More than 560 interactions between 79 cellular proteins and NS1 and NS2 proteins from 9 different influenza virus strains have been identified. A yeast two-hybrid screening strategy was used to identify host factors supporting NS1 and NS2 functions. NS1 is a multifunctional protein and a virulence factor while NS2 is involved in nuclear export of viral ribonucleoprotein complexes. Influenza A NS1 and NS2 proteins are encoded by the RNA segment 8 of the viral genome.
